Life Sciences and Pharmaceutical Analysis
EMILIE allows the characterization of pharmaceutical samples available in very small quantities such as biopharmaceuticals, oligonucleotides, proteins, and other nanomedicines, paving the way for breakthroughs in drug development and quality control.
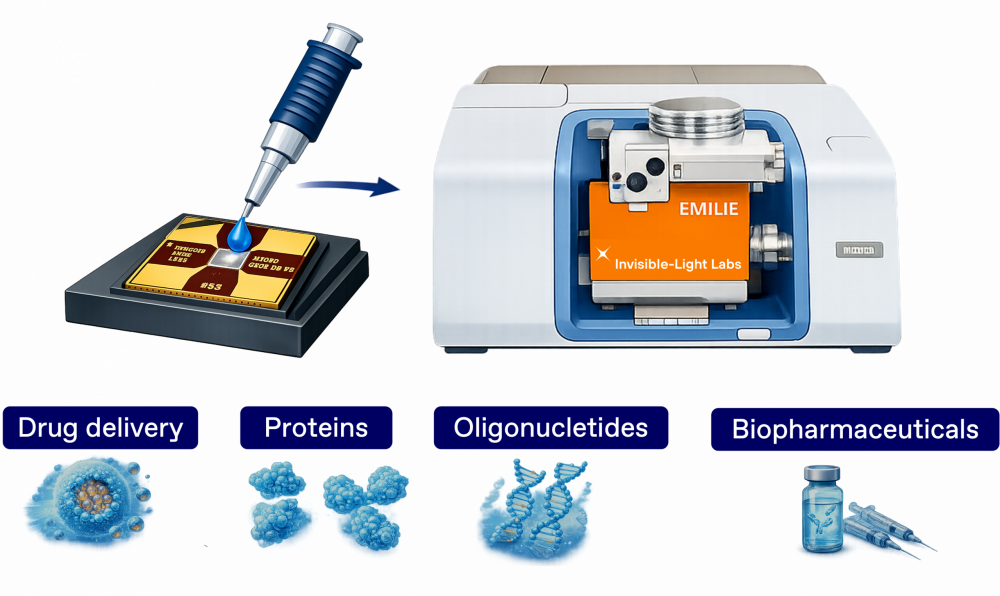
Characteriation of biopharmaceutical samples and their contaminants
Be it for drug development or quality control, EMILIE provides a simple and sensitive way to chemically characterize samples available in small quantities.
Read More
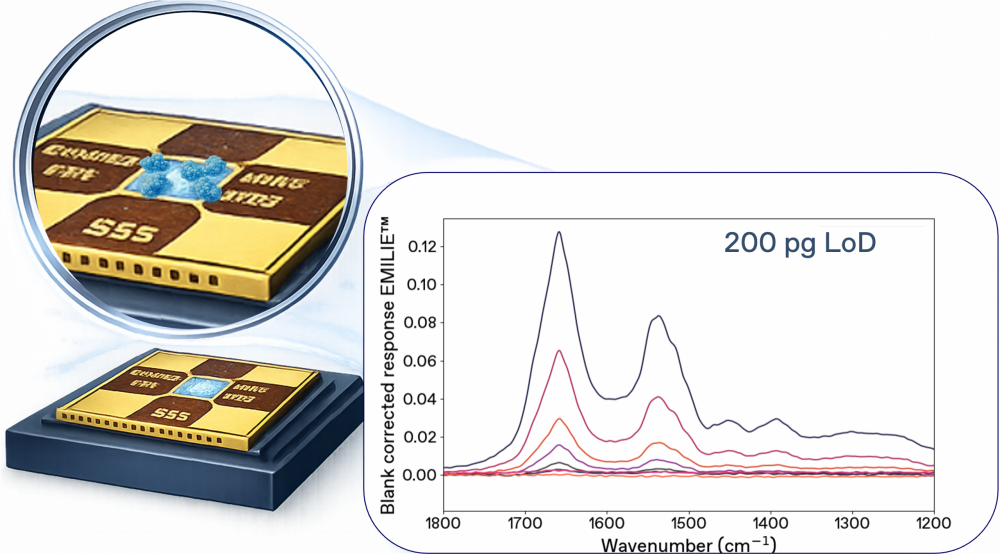
Chemical characterization of proteins by NEMS-FTIR with EMILIE
After deposition of the sample and drying, the EMILIE nanomechanical sampling and sensing chip is transfered directly to the EMILIE infrared analyzer for NEMS-FTIR analysis.
EMILIE in compared to the state-of-the-art pharmaceutical characterization
200pg
detection limit for proteins
0x
water bands obscuring amide peaks thanks to measurements under vacuum
4x
Complementary orthogonal techniques for further analysis: RAMAN spectroscopy, SEM/EDX, O-PTIR, and LDIR
0x
labels necessary
EMILIE
The first nanoelectromechanical analyzer for FTIR analysis
Deposit any non-volatile sample directly on the EMILIE nanomechanical chips and obtain chemical information in minutes!

Request a quote!
